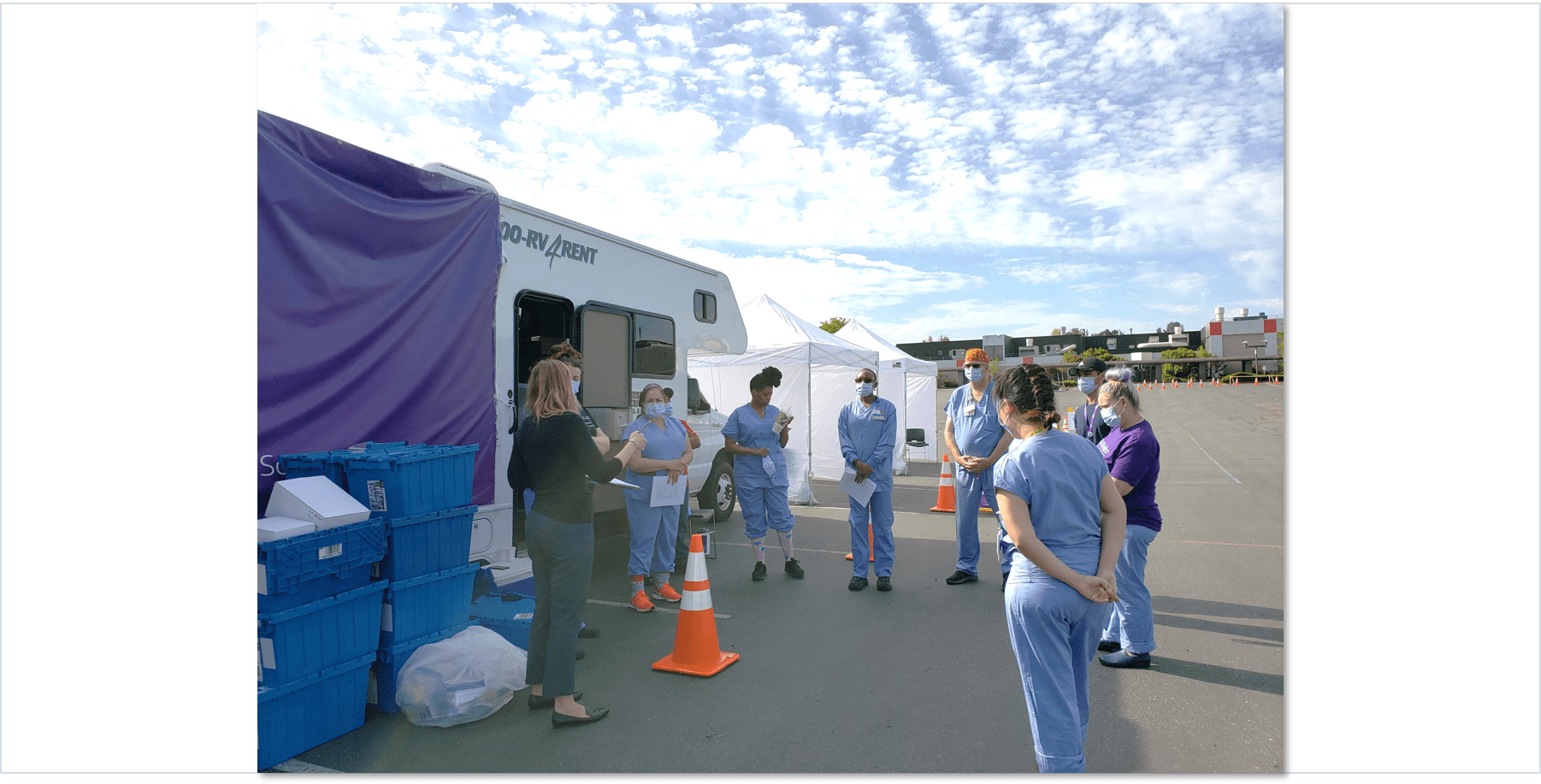
As of the end of June, there have been more than 10 million confirmed COVID-19 infections world-wide, with a quarter of those in the United States. The World Health Organization Director-General Tedros Adhanom Ghebreyesus predicted that “the worst is yet to come,” and the solution remains the same since the beginning of the pandemic – “test, trace, isolate, and quarantine.” Safe and efficient testing of sufficient levels of the population is essential to support mitigation and ideally suppression of the virus to levels low enough to safely open public life again. A significant portion of SARS-CoV2 testing occurs in off-site testing centers set up by medical facilities, public health departments, and private organizations.
To support off-site COVID-19 testing centers (OSCTC) in this period of rapid change, the Network for Regional Healthcare Improvement (NRHI) and NRHI member and research partner, Qualidigm, have been leading a national effort since March to identify and disseminate considerations for off-site testing.
Our current Phase II work follows a Phase I 30-day rapid assessment from mid-March to April, producing preliminary considerations for off-site testing. These considerations were based on a small sample of eleven qualitative interviews with regional leaders of OSCTCs affiliated with hospitals or health systems and focused on five themes:
- Leaders and partners
- Staff protection
- Testing capacity and follow-up process for results
- Patients demographics
- Test reimbursement.
A summary of the findings has been published in Healthcare: The Journal of Delivery Science and Innovation and the full report has been released. The major considerations have been organized in an easy-to-follow infographic and disseminated broadly to support sites looking to establish or improve testing centers.
Ongoing research is imperative as testing demand increases, guidance on priority populations emerges, testing types and supplies evolve, and federal support for testing is reduced. With the current surge of new cases and the ominous predictions for the fall and winter, the nation needs to be able to test safely, efficiently, and sufficiently.
Our Phase II research includes a longitudinal analysis of sites described in Phase I, as well as interviews with public health departments, sites carrying out testing in highly impacted locations (jails and long term care facilities), those serving vulnerable populations, high risk workplaces, and private testing sites such as pharmacies.
Some of our key research questions for this phase address access to testing among vulnerable populations, as well as the alignment of OSCTCs with public health entities, community-based organizations, and other health systems to effectively address the testing needs, limitations, and considerations across the population. Considerations will be updated to include emerging themes, such as coverage and reimbursement, determining which type of test to use, and considerations for workplace testing.
In an interview Dr. Dustin Calhoun, University of Cincinnati Associate Professor of Emergency Medicine and a clinical advisor to this research, described the benefit of this work:
“Everyone who does this is finding their own little areas of differences and their own problems to overcome but everyone is coming up with a way to overcome them. So rather than continuously reinventing the wheel, let’s gather what are those big problems and not only that, but how did others overcome them – what are the keys to opening those doors. Any resource that isn’t being used to try to figure how to solve a problem, can be used to actually perform the function we are trying to do. So every problem we have to individually overcome is one less resource being used toward actual testing. Collective benefit from this experience is valuable.”
View the full interview with Dr. Calhoun below.
The research findings will be submitted to a peer-reviewed journal for publication and accompanied by an online toolkit to support broad dissemination of our findings and facilitate adoption of promising practices.
Share what has worked well for your off-site testing center with others. Please, reach out to us at